Ammonia Testing for Water Quality
Good water quality within any aquarium is essential to the long-term health of all aquatic species. Poor water quality is the main factor leading to poor health, disease and even fish death. The only way to determine water quality is through accurate testing. Following feeding, ammonia is produced from the waste of fish and invertebrates, most of the excreted nitrogen is in the form of ammonia released directly from the fish through their gills. Even at relatively low concentration, ammonia within an aquatic environment is highly toxic and may cause gill damage, skin irritation and sometimes death. Therefore, the testing of ammonia levels within a tank is essential.
Physical signs of fish with acute ammonia poisoning include damaged gills, clamped fins, unusual darkened areas on the body, gasping at the surface, and cellular damage often seen as ragged fins. If left unattended the fish will slowly become lethargic and either succumb to disease or inability to osmoregulate.
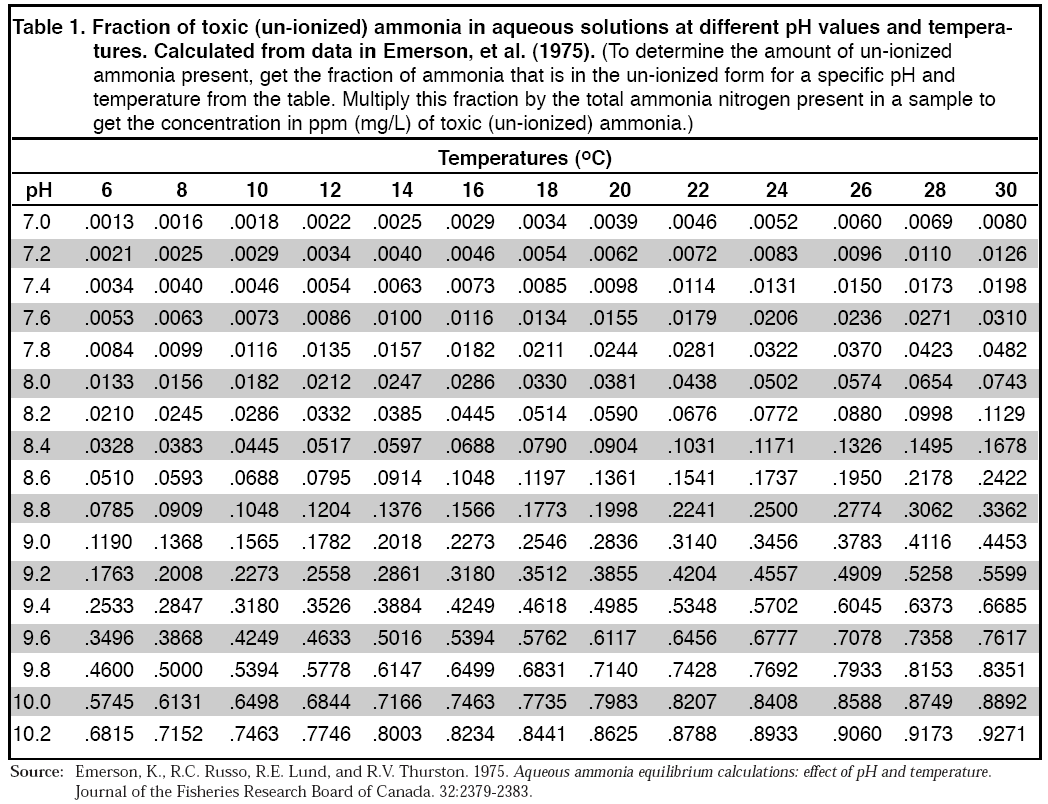
Ammonia exists in two forms, namely, unionized (NH3) and ionized (NH4+). Both forms are measured together and are referred to as total ammonia nitrogen (TAN). Most ammonia test kits are actually testing TAN (Total Ammonia Nitrogen). TAN is a combination of unionized ammonia (NH3) and ionized ammonium (NH4+). Ammonia (NH3) is a highly toxic chemical to fish and changes from ammonia to ammonium and back again relative to the pH level and temperature. API Ammonia Test Kits, both Liquid and Dip Strip, test for TAN. To calculate the unionized ammonia (NH3) from TAN you can use the table at the end of this article. However, when ammonia is present even in trace amounts your fish are being damaged. Regular testing and taking corrective action immediately are advised to protect your fish. Once fish have started to die from ammonia poisoning it is very difficult to make corrections without further loss of fish. When testing your aquarium water, the only safe level for ammonia is zero.
Unlike nature, an aquarium is a closed environment, where all the ammonia and ammonium from the wastes excreted from the fish, uneaten food, and decaying plants stays inside the aquarium. Fish feces and urine, as well as any uneaten food, are quickly broken down into either ionized or unionized ammonia. The ionized form, Ammonium (NH4+) is predominant if the pH is below 7 and is relatively non-toxic to fish. The unionized form, Ammonia (NH3), is predominant if the pH is 7 or above, and is highly toxic to fish. The problem is that slight changes in pH can take an environment of low toxicity to high toxicity. Remember, even low toxic conditions or slight toxic conditions cause damage to fish and lower the immune systems of fish. Even at low levels, ammonia problems are documented in reduced growth and damage to gill filament tissue. The lethal effects of exposure to ammonia are severe gill damage leading to suffocation, kidney damage due to inability to osmoregulate, and the increased inability to secrete ammonia from the body resulting in metabolic and physiological imbalance. Temperature also plays an important part along with pH in the toxicity level of ammonia.
A lethal poisoning level for the unionized (NH3) form is 1.00mg/l; a sub-lethal level is 0.05mg/l (Noga, E.J., (1996) Fish Disease, diagnosis and treatment. p 62-66.). The toxicity of ammonia is dependent on the water pH, and temperature since an increase in pH and temperature both favor the conversion of NH4 to NH3. The percentage concentration of NH3 in water can be determined from a standards chart (Appendix 1).
The levels of ammonia are reduced by biological filtration and in a closed system, such as the aquarium, should be zero. The biological filtration houses beneficial nitrifying bacteria to break-down ammonia produced by fish to nitrite and then nitrites are converted to nitrates. This is commonly called the “Nitrogen Cycle”, see diagram above. The design of the system, limiting the fish load to a realistic carry capacity based on testing, and a suitable feeding regimen are the best ways to control ammonia.
In conclusion, attempting to educate the average hobbyist on the use of the chart below is problematic at best. It provides a false sense of safety when they think that ammonium is not harmful, which is not true. Ammonium and ammonia are released from the fish, mostly from their gills (85% and up), the remaining accumulation of ammonia and ammonium in the aquarium come from feces, urine, uneaten fish food and other amine organics. If the ammonia or the ammonium is high in the aquarium it is harder for the fish to release either of these compounds from their blood. This can cause the ammonia/ammonium levels in the fish to be higher than the surrounding aquarium water being tested.
The only safe TAN (Total Ammonia Nitrogen) level for an aquarium is zero. API AMMONIA TEST KIT provides the answer for the TAN (total ammonia nitrogen) in a responsible way for all hobbyists to easily understand and, most importantly, to protect their fish!
